
Associate Professor
Academic History
- B.Sc. (1995) University of British Columbia;
- Ph.D. (1999) Dalhousie University, Halifax, NS;
- NSERC/CIHR/MSFHR Postdoctoral Fellow (1999-2003) University of British Columbia
Contact Information:
- 604-822-1630
Research Interests
Our research strives to understand genome evolution in single-celled eukaryotes, focusing on changes in genome size. As genomes are reduced in size, there are likely to be changes in the amounts of non-coding material. Of particular interest is the retention, reduction and/or removal of introns. Introns interrupt genes, and are removed from mRNA by a large macromolecular protein and RNA complex called the spliceosome. In metazoans, where genomes tend to be quite large, introns can be quite long (sometimes several kb), and the spliceosome is extremely large with over a hundred proteins. In contrast, the yeast Saccharomyces possesses a much smaller genome and has relatively few introns that tend to be short. The yeast spliceosome is also predicted to be smaller, with only ~80 proteins. A lineage of lesser-studied eukaryotes, the microsporidia, are distantly related to fungi and possess genomes that are very tiny; at the extreme, smaller than many bacterial genomes. Using microsporidia as a model system, we are examining the effects of genome reduction on the evolution of introns and the spliceosomal machinery.
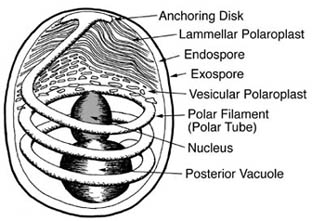
Microsporidia are a large group of intracellular parasites. Outside of host cells they exist as highly resistant spores, which possess organelles almost entirely relegated to their amazing process of infection. The most prominent of these is the polar filament that is wrapped around the spore contents. With the appropriate environmental triggers, the filament is rapidly everted, breaking through the spore wall and forming a projectile tube that can pierce a nearby cell, much like a harpoon. The infective spore contents can then pass through the tube and are injected into the host cell. The parasite grows and divides within the host’s cytoplasm, eventually producing more spores and killing the host cell.
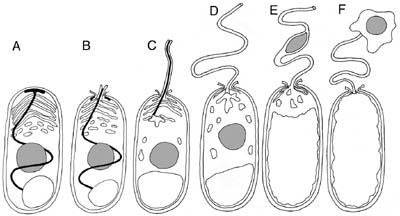
Using molecular techniques, our research strives to address genome evolution in these unusual eukaryotes. Currently we are focusing on three general areas of research: (1) A molecular examination of intron positions and lengths among microsporidia possessing a range of genome sizes.
(2) A largely bioinformatic assessment of the differing spliceosomal components among microsporidia, with comparisons to more distantly related fungi.
(3) Small-scale molecular sequence surveys of microsporidia possessing different genome sizes.
Team Members
Cameron Grisdale, PhD student
Alex Ardila-Garcia, PhD student
Selected Publications
Lee, R.C.H., S.W. Roy and N.M. Fast. 2010. Constrained intron structures in a microsporidian. Mol. Biol. Evol. (in press; doi: 10.1093/molbev/msq087)
Gill, E.E., R.C.H. Lee, N. Corradi, C.J. Grisdale, V.O. Limpright, P.J. Keeling and N.M. Fast. 2010. Splicing and transcription differ between spore and intracellular life stages in the parasitic microsporidia. Mol. Biol. Evol. 27:1579-1584.
Gill, E.E., J.J. Becnel and N.M. Fast. 2008. ESTs from the microsporidian Edhazardia aedis. BMC Genomics 9:296.
Williams*, B.A.P., R.C.H. Lee*, J.J. Becnel, L.M. Weiss, N.M. Fast and P.J. Keeling. 2008. Genome reduction and expansion in microsporidia. BMC Genomics 9:200.
Lee, R.C.H., B.A.P. Williams, A.M.V. Brown, M.L. Adamson, P.J. Keeling and N.M. Fast. 2008. Alpha and beta tubulin phylogenies suggest a close relationship between the microsporidia Brachiola algerae and Antonospora locustae. J. Eukaryot. Microbiol. 55:388-392.
Gill, E.E and N.M. Fast. 2007. Stripped down DNA repair in a highly reduced parasite. BMC Mol. Biol. 8:24 (p.1-14)
Gill, E.E. and N.M. Fast. 2006. Assessing the microsporidia-fungi relationship: combined phylogenetic analysis of eight genes. Gene 375:103-109.
Williams, B.A.P., C.H. Slamovits, N.J. Patron, N.M. Fast, and P.J. Keeling. 2005. A high frequency of overlapping gene expression in compacted eukaryotic genomes. Proc. Natl. Acad. Sci. USA 102:10936-10941.
Keeling, P.J., B.A.P. Williams, J. Law, N.M. Fast, and C.H. Slamovits. 2005. Comparative genomics of microsporidia. Folia Parasitol .52:8-14.
Keeling, P J., J.M. Archibald, N.M. Fast, and J.D. Palmer. 2004. The evolution of modern eukaryotic phytoplankton.Science , 306:2191.
Slamovits, C., N.M. Fast, J.S. Law, and P.J. Keeling. 2004. Genome compaction and stability in microsporidian intracellular parasites. Curr. Biol. 14:891-896.






